Broader and deeper protein content for optimized discovery potential
Accelerate your research in oncology, neurology, cardiology, infectious disease, and more.
Download our Tech Note to learn how the SomaScan 11K Assay works
The SomaScan® 11K Assay:
Your access to half of the human proteome
The SomaScan Assay is the only proteomic technology capable of measuring rapidly (high throughput), broadly (thousands of proteins simultaneously), and deeply (high-and low-abundance proteins).
This industry-leading multiplex platform provides 11,000 total protein measurements, including 10,000 unique human proteins measured. That’s half the human proteome.
What does this mean for your research?
The study of proteomics is essential for the discovery of novel bimarkers and potentially causative mechanisms. Acquiring such robust data on numerous proteins at once offers groundbreaking insights into predicting response, mechanisms of action and biomarker translation – and with the confidence that you are seeing it all.
The broadest, deepest disease coverage
The SomaScan 11,000-plex menu is the most powerful proteomics solution today, enabling researchers to decipher human biology by casting the widest net possible.
Benefit from a high-plex, sensitive, and reproducible proteomic tool designed for drug discovery, pre-clinical, and clinical drug development across a wide range of diseases and conditions.
Do you need to compare data from SomaScan 7K or 5K projects with the new SomaScan 11K platform? Or are you interested in learning more about what the 11k assay can do for your research? Maximize your data’s potential with our comprehensive tools that allow you to seamlessly compare data across assay versions and eliminate the need to run additional samples for data bridging.
The comprehensive coverage of a broad selection of protein targets is mapped to biological processes and various disease areas, shown at right.
Detect proteins across every major biological pathway
% Proteomic Coverage by Platform for Selected Pathways
Our precision is backed by orthogonal validation
From our original 1,300-plex menu to today, we have maintained consistent quality metrics for specificity, sensitivity, reproducibility, and more.
Our customers rely on consistent quality, so they know their data is always accurate and meaningful. To meet their needs, we provide the largest number of protein measurements and the greatest number of orthogonally confirmed protein reagents in the proteomics industry.
Primary validation of all SOMAmer® Reagents includes:
- Determination of equilibrium binding affinity dissociation constant
- Pull down assay of cognate protein from buffer
- Demonstration of buffer dose response in the SomaScan Assay
- Estimation of endogenous cognate protein signals in plasma
- Required for inclusion in the SomaScan Assay
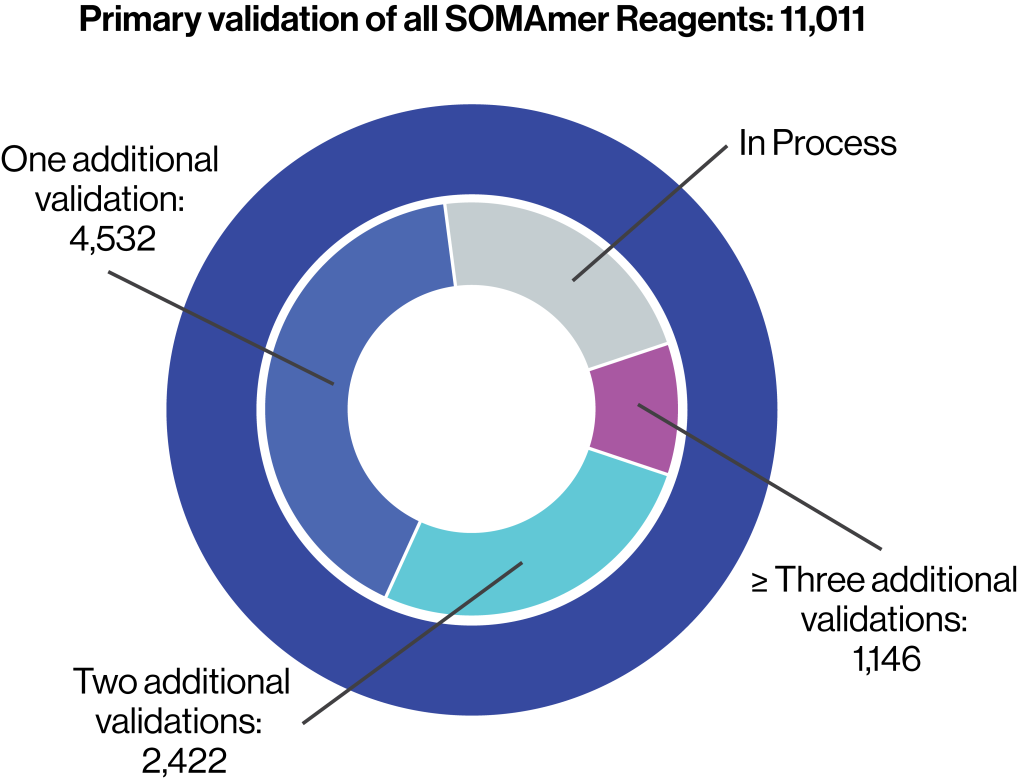
Over 8,000 SOMAmer Reagents have at least one additional form of orthogonal validation
Over 3,000 SOMAmer Reagents have undergone two or more forms of orthogonal validation

Let us help you maximize your data’s potential with our comprehensive suite of tools.
11K Platform Bridging
With some assay platforms, integrating data across versions can be difficult and time consuming. When you use the SomaScan platform, we make it easy for you to compare your 11K data to your 7K and 5K data.
Have confidence in data integration across SomaScan platforms
We’ve developed and qualified the method that allows you to quickly compare data from projects run across our SomaScan 5K, SomaScan 7K, and new SomaScan 11K platforms.
Save time and money
With the SomaScan platform, we have developed simple solutions to compare 11K data to your 7K or 5K data for Human EDTA Plasma and Serum. This means your project may not require additional samples if you ever need to compare your project to a previous platform project.
Maximize your insights across long term studies
Our accessible, open-source tools break down data silos and unlock deeper insights across long term studies that span different versions of the SomaScan platform.
SomaScan Assay services can be found at: