How Blood Proteomics Is Accelerating Neuro Drug Discovery
How Blood Proteomics Is Accelerating Neuro Drug Discovery
Neurodegenerative diseases affect more than 57 million people globally, a figure expected to double within two decades1,2. This alarming projection reflects three major clinical challenges: difficulty diagnosing disease in early, treatable stages; a lack of curative therapies; and limited treatment options2,3. Gradual onset, overlapping symptoms and biological heterogeneity complicate both diagnosis and therapeutic development. In response, blood-based biomarkers have emerged as a promising solution, offering a more accessible, scalable way to detect, monitor, and stratify disease4.
The translational promise of blood-based biomarkers
Compared to cerebrospinal fluid (CSF) sampling or neuroimaging, blood tests are minimally invasive and cost-effective. The FDA’s 2025 approval of the first blood-based in vitro diagnostic for Alzheimer’s disease (AD) highlights growing confidence in the circulating proteome as a clinically informative matrix5.
Accelerating this shift toward investigating the plasma proteome, the Global Neurodegeneration Proteomics Consortium (GNPC) released one of the most harmonized proteomics datasets to date2. Publicly released in July 2025, the GNPC Version 1 (V1) dataset comprises more than 250 million protein measurements generated from serum, plasma and CSF samples collected from 18,645 individuals across AD, Parkinson’s disease (PD), frontotemporal dementia (FTD) and amyotrophic lateral sclerosis (ALS).
In the Nature Medicine publication accompanying the GNPC V1 release, the authors present three use cases demonstrating how plasma proteomics can interrogate key biological questions in neurodegenerative and aging research2. Using the GNPC dataset, they show that plasma proteomic profiles:
- Indicate both disease status and severity, showing strong associations with disease type and clinical progression;
- Reflect disease-specific patterns of premature aging, based on plasma proteomic organ aging models;
- Correlate with APOE ε4 carrier status, helping distinguish AD-specific effects from APOE genotype-related pathways.
Scalable discovery: Profiling 10,000+ proteins from just 55 μL of plasma
To support high-resolution plasma proteomic analysis, GNPC selected the SomaScan™ Assay as the primary platform (Figure 1) 2. With unmatched breadth, the latest SomaScan™ 11K Assay simultaneously measures over 10,000 human proteins – including low-abundance proteins – without the need for depletion or enrichment.
Unlike other affinity-based platforms, it does not require bridging samples across batches or sites, streamlining workflows and data analysis for multi-center and longitudinal clinical trials. In a recent independent comparison of eight proteomics platforms, the SomaScan Assay demonstrated the highest analytical precision and the most comprehensive coverage of FDA-approved plasma protein biomarkers6.
Antibodies: The “hidden” 20% of the plasma proteome
Comprising roughly 20% of the plasma proteome by mass, antibodies play critical roles in disease pathology and treatment response. Their stability over time and sensitivity to physiological changes make them ideal candidates for longitudinal biomarkers and pharmacodynamic monitoring. Yet traditional proteomics methods offer limited access to antibody–antigen interactions at scale.
The KREX™ Assay addresses this gap with highly multiplexed autoantibody (AAb) profiling against more than 1,800 full-length, properly folded human proteins. This enables biologically relevant antibody–antigen binding, revealing which proteins the immune system flags as “abnormal” and how it responds to disease or therapy. These insights help uncover early immune dysregulation, enable patient stratification and inform immune-targeted therapeutic strategies.
Selected studies illustrating the translational value of AAb profiling in neurodegeneration include:
- Potential early diagnostic biomarkers: Six AAbs elevated in patients with mild AD compared to non-AD dementia, several targeting proteins involved in cognitive decline and Tau phosphorylation7.
- Potential immune-targeted intervention biomarkers: Eight AAbs elevated in PD patients seropositive for Helicobacter pylori, supporting epidemiological links between the pathogen and worsened motor symptoms8.
A new era for neurodegenerative research
As pharmaceutical pipelines shift toward precision and predictive biomarkers, high-plex plasma proteomics is uncovering real-time, actionable insights into disease biology. While this article focused on neurodegeneration, circulating molecular signatures are increasingly recognized as valuable across a broader spectrum of CNS disorders, including autism spectrum disorder and psychiatric conditions.
Standard BioTools™ supports translational and clinical research through scalable services for SomaScan and KREX Assays. By delivering high-quality, reproducible data from small sample volumes, we help pharmaceutical teams like yours advance biomarker-driven strategies – from bench to bedside.
References
- Giri, P.M. et al. “Neuroinflammation in neurodegenerative disorders: Current knowledge and therapeutic implications.” International Journal of Molecular Sciences 25 (2024): 3995.
- Imam, Farhad et al. “The Global Neurodegeneration Proteomics Consortium: biomarker and drug target discovery for common neurodegenerative diseases and aging.” Nature medicine vol. 31,8 (2025): 2556-2566.
- Bisi, N. et al. “Early diagnosis of neurodegenerative diseases: What has been undertaken to promote the transition from PET to fluorescence tracers.” Molecules 29 (2024): 722.
- Alcolea, D. et al. “Blood biomarkers in neurodegenerative diseases: Implications for the clinical neurologist.” Neurology 101 (2023): 172–180.
- S. Food and Drug Administration. “FDA Clears First Blood Test Used in Diagnosing Alzheimer’s Disease.” (2025): fda.gov/news-events/press-announcements/fda-clears-first-blood-test-used-diagnosing-alzheimers-disease.
- Kirsher, Douglas Y et al. “Current landscape of plasma proteomics from technical innovations to biological insights and biomarker discovery.” Communications chemistry 8,1 279. 25 Sep. 2025.
- Wang, B.Z. et al. “Identification of novel candidate autoantibodies in Alzheimer’s disease.” European Journal of Neurology 27 (2020): 2,292–2,296.
- Suwarnalata, G. et al. “Augmentation of autoantibodies by Helicobacter pylori in Parkinson’s disease patients may be linked to greater severity.” PLoS One 11 (2016): e0153725.
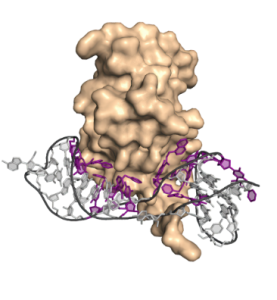
Figure 1. The SomaScan Assay utilizes proprietary DNA aptamers (SOMAmers™) to bind proteins based on their unique three-dimensional structures.
More blogs
BlogAntibodies: The Hidden 20% of the Plasma Proteome
Antibodies make up 20% of plasma yet most proteomics studies overlook their specificity and early disease signals.
BlogHigh-Plex Affinity Proteomics for Plasma
High-plex affinity proteomics enables deep, reproducible plasma biomarker discovery. See how the SomaScan 11K Assay delivers scale, sensitivity, and coverage.
BlogThe Challenges of Plasma Proteomics for Biomarker Discovery
Why is protein biomarker discovery in plasma so difficult? Explore how protein complexity and dynamic range limit detection of low-abundance signals.





